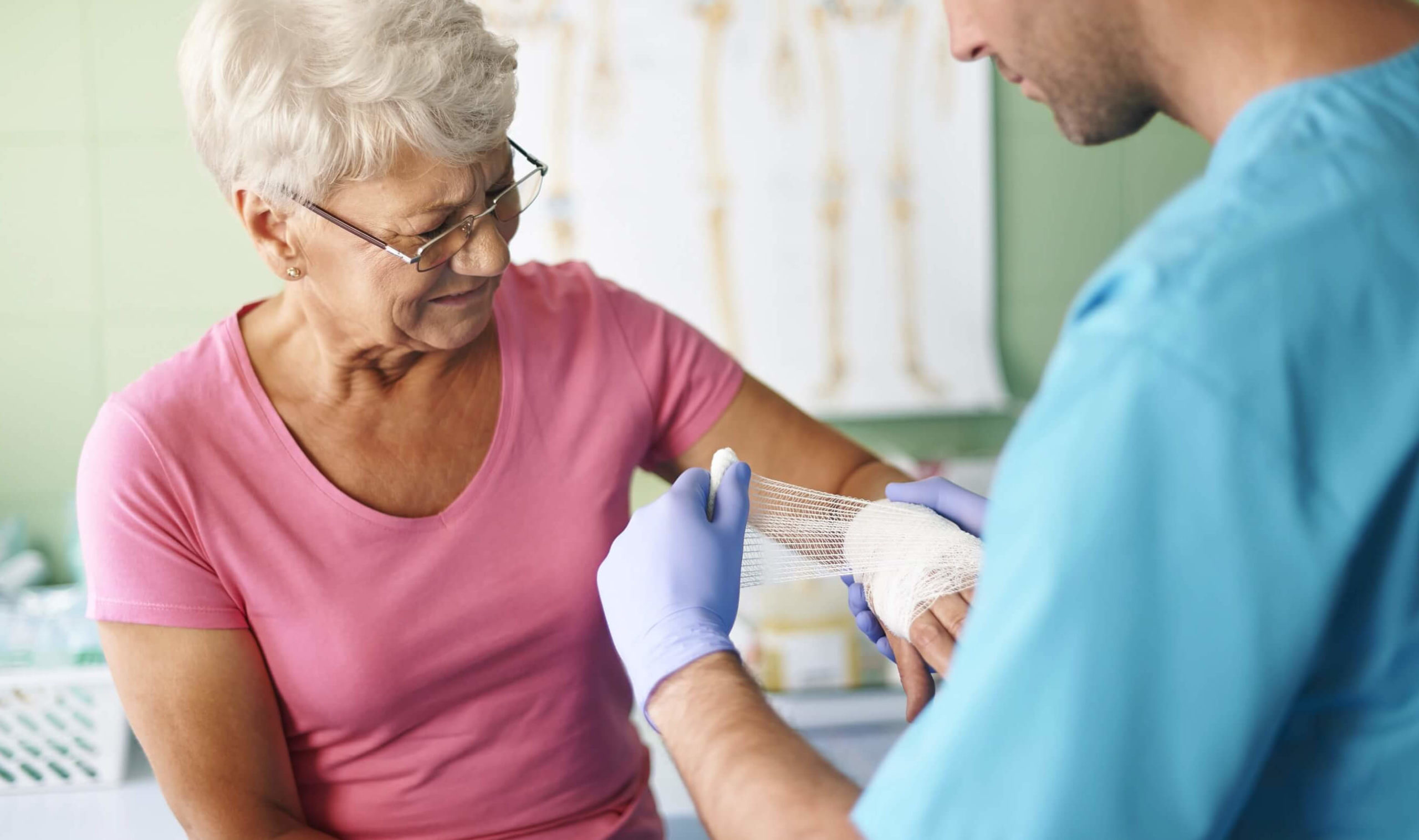
EPIEN Medical’s proprietary debridement process is called Desiccation Shock Technology (“DST”) .
Technology Platforms
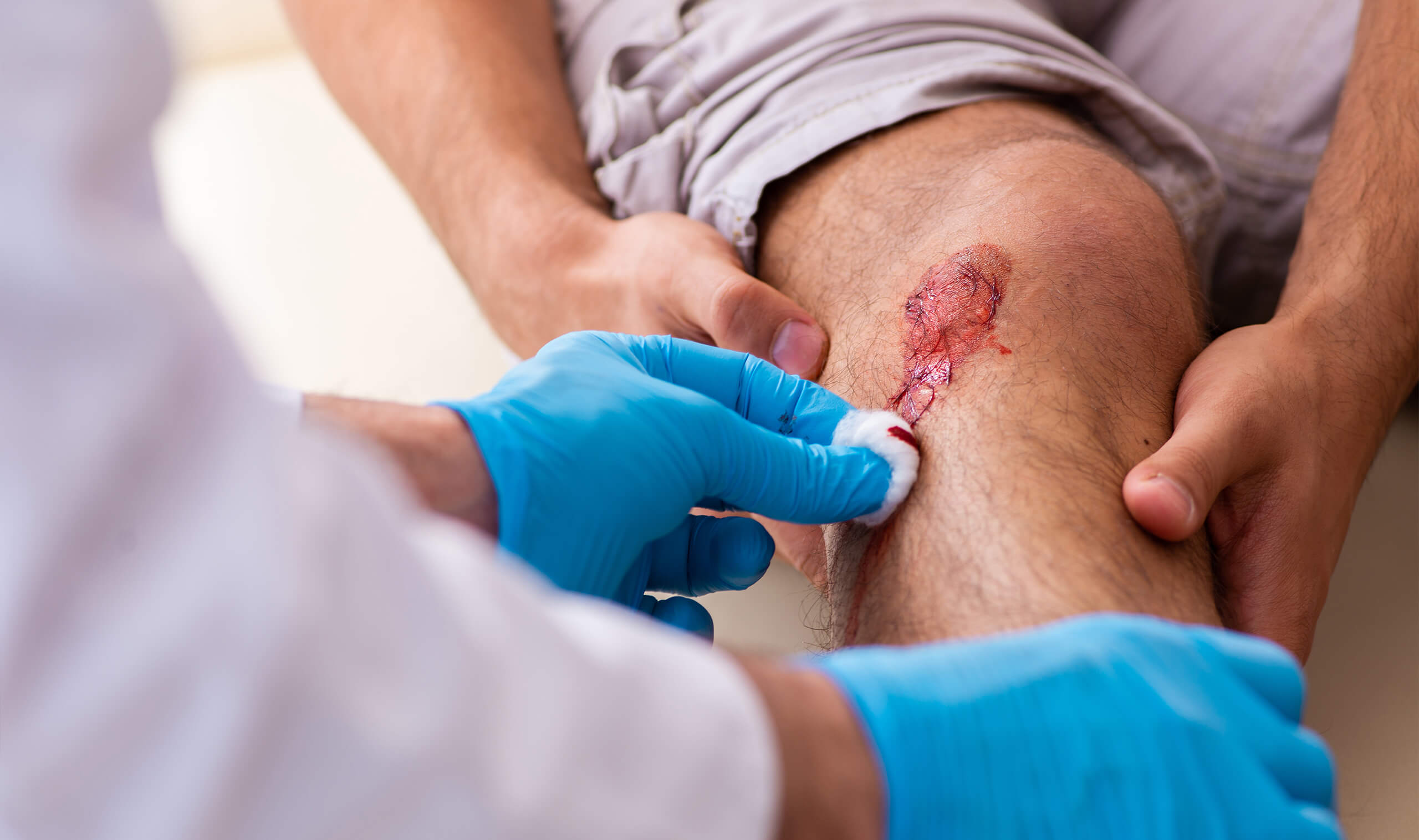
DST is a novel breakthrough for treating epidermal and dermal tissue injuries.
This is the foundation for a distinct new class of aggressive chemical formulations whose primary purpose is to totally eradicate biofilm from infected tissue surfaces. EMI’s goal in the development of DST has been to build a technology platform that could quickly deliver an intense molecular cleansing action through a means of desiccation that leaves any wound microorganism-free. Once the wound is free of any bacteria and infectious infiltrate, the wound is reset to allow the body’s own natural defense mechanisms to resume its healing protocol.
Our DST works to remove infected tissues through two processes. The first is by absorbing so much water from the infected area (desiccation) that the damaged tissue on the surface becomes dried, shrinks, and starts to pull away from the healthy tissue below. As a result, the infectious material is destroyed. The second process is a superior mechanical rinsing action. The DST liquids are very dense and viscous. For that reason, they are very effective at mechanically dislodging surface debris and trapping it so it can be rinsed away more easily. These two processes occur at the same time during DST product application. This allows practitioners to completely clean and sanitize an infected site anywhere on the body surface in less than 60 seconds without harm to adjacent healthy tissues. The products are simply delivered onto tissues with a syringe or other medical applicator. It is then removed easily by rinsing with water or saline.